Research
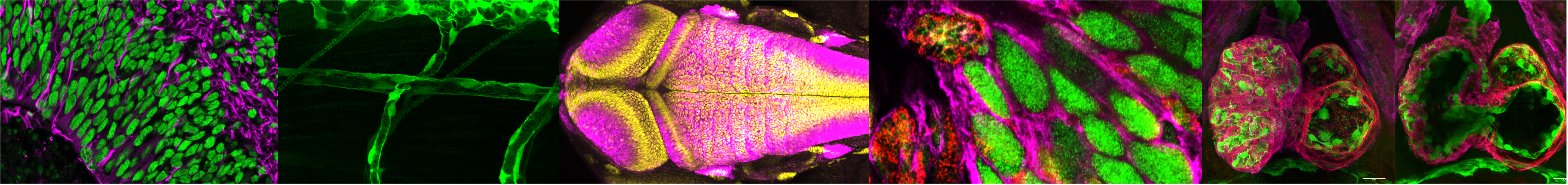
Our lab investigates how haploinsufficiency of chromatin regulators—the histone methyltransferase KMT2D and the histone acetyltransferases KAT6A and KAT6B—disrupts progenitor cell fate in rare pediatric epigenetic disorders. We test whether chromatin regulators control a conserved developmental timing program across cardiac and neural fate transitions, using zebrafish and human iPSC models.
Neural Progenitor Timing
Understanding how KMT2D deficiency causes premature but unstable activation of lineage transcription factors and accelerated G1 accumulation in neural progenitors.
Cardiovascular Development
Investigating KMT2D's role in Notch-mediated cardiovascular patterning, endothelial tip/stalk cell selection during sprouting angiogenesis, and ontogeny-specific pericyte histone landscapes.
Conserved Timing Mechanisms
Testing whether disrupted developmental timing is a shared pathogenic mechanism across KMT2D, KAT6A/B, and related chromatinopathies using developmental time courses and cross-species functional rescue.
EpiFlow Technology
Developing high-throughput spectral flow cytometry platforms that multiplex histone H3 modifications with cell cycle and lineage markers at single-cell resolution to detect subtle chromatin changes in heterogeneous populations.
Zebrafish Models
Using the kmt2dzy59 null line and zebrafish models to validate in vivo developmental timing signatures and test functional rescue strategies for Kabuki syndrome.
Patient iPSC Biobank
Maintaining a centralized biobank of KAT6A/B patient-derived iPSC lines in partnership with the KAT6 Foundation.